Past Research
Thermal Management of High Heat Flux Electronics
Caleb Anderson, Nicole Cassada, Torben Grumstrup, Taylor Bevis, Bryan Burk, Jensen Hoke, Josh Richey
Partners: Lawrence Livermore National Laboratory (LLNL), Office of Naval Research (ONR), DEVCOM Army Research Laboratory (ARL), Air Force Research Laboratory (AFRL)
As electronics trend towards increased miniaturization and greater performance, the need for high-performance cooling of becomes critical. The resulting heat fluxes become extremely high and require more complex cooling techniques. Heat generated by high power density devices such as laser diode bars and arrays, power electronics, and other semiconductor switching electronics must be efficiently removed from the device to enable optimal functionality and avoid catastrophic overheating. Microchannel heat exchangers are an attractive solution to this problem. This style of device utilize advanced micromachining techniques to fabricate low thermal resistance cold plates with small hydraulic diameter and large aspect ratio channels. Microchannel cold plates operating under two-phase flow boiling conditions (microchannel evaporators) are an attractive thermal management strategy for cooling high heat fluxes (>1 kW/cm-2) with nearly isothermal flow conditions and at lower pumping powers compared to single-phase cooling solutions. However, two-phase flow is complex and is susceptible to a variety of flow instabilities.
A previous partnership with Lawrence Livermore National Lab (LLNL) developed low thermal resistance microchannel ( µm) evaporators out of thin (0.3 – 0.5 mm) silicon wafers using a deep reactive ion etching technique. The goal of the project was to improve the optical power density of laser diode arrays by employing microchannel evaporators to increase the output power of the devices and reduce the pitch between adjacent bars in an array. The devices have a 1 mm x 10 mm platinum strip heater on the backside which acts as a surrogate laser diode bar. A custom two-phase pumped refrigerant loop was built to test these microchannel cold plates under two-phase cooling with R134a. Experiments have shown that up to 150 W and 1.5 kW/cm2 can be successfully dissipated with these devices, depending on channel and fin geometries and fluid conditions. The data collected in this work was used to valid microchannel heat transfer models. Accurate modeling of microchannel evaporator performance is necessary to predict and design practical devices using this thermal management technique. A finite element analysis (FEA) conjugate heat transfer model was created in COMSOL Multiphysics to compare the predictions of a variety of two-phase heat transfer correlations with the experimental results. The model coupled two-phase pressure-drop calculations from literature correlations with locally calculated fluid properties and locally applied heat transfer coefficients as calculated from the heat transfer correlations.
Following the conclusion of the LLNL project, the team received funding from the Office of Naval Research (ONR) to assist the DEVCOM Army Research Lab (ARL) and Air Force Research Lab (AFRL) on a large-scale collaborative effort. The goal of the project was to improve the Size, Weight, and Performance (SWaP) of pulsed-power systems operating with two-phase thermal management systems by implementing a variety of controls, modeling, and improved implementation strategies. One of the evaporators which was previously tested under steady state conditions was tested under transient heat loads. During the transient heat load testing, vapor backflow (as shown in the white dotted traces) at the onset of boiling was unexpectedly observed despite the presence of the stabilizing inlet restrictions due to the large wall superheat temperatures above the local fluid temperature. These flow instabilities and temperature overshoots could be seriously detrimental to a real device operating under transient heat loads with such an evaporator. Thus, research into developing a predictive model and a means of reducing the peak temperatures and vapor backflow is currently ongoing.
A new project funded by LLNL is currently underway towards improving the performance and optical power density of laser diode arrays with a novel microchannel cooling architecture. A scanning electron microscopy image shows a sample of the large aspect ratio microchannels which can be fabricated for the current research. Since laser diode arrays function with large power densities and are closely packed together, the resulting heat fluxes are typically in excess of 1 kW/cm2 and benefit from the highly efficient cooling mechanisms of microchannel cold plates.
Thermal Management of Power Electronics Packages
Past: Justin Hollis Partners: Army Research Labroatory (ARL)
Demand for more versatile, higher power, longer range, and more capable electric vehicles is rapidly growing. To meet these needs, electric vehicles will require more power to enable use in various industries: from commercial transportation and shipping to military vehicles of multiple sizes and capabilities. Electric vehicles are reliant on power electronics packages to manage onboard power systems. Improving the performance, reliability, and cost of power electronics packages is critical to the development of more robust and cost-effective electric vehicles. The reliability of power electronics can be improved by reducing the maximum temperature and smoothing out temperature fluctuations that the package experiences during regular operation. To meet both of these goals, phase change materials (PCMs) can be incorporated into the power electronics packaged to dissipate heat during transient operation. In this work, multiple PCM and PCM composites (Erythritol, Erythritol and nickel, Erythritol-copper composite, and Indium) were simulated in Parapower for both single-sided and dual-sided package configurations to quantify the impact on peak junction temperature and temperature variability during real drive cycles for on-road vehicles. The Erythritol-copper PCM in a single-sided package with 24 die reduced the maximum junction temperature by 26°C, from 151°C to 125°C, while only increasing the volume of the package by 20%. The dual-sided package reduced peak temperature by 6°C, but was not as effective as the single-side package due to geometrical placement constraints and thermal pathways. Both single and dual-sided PCM packages decreased the temperature fluctuations in the power electronics package, which help to improve reliability and durability.
Distributed Power and CO2 Sequestration Reformer-Engine
Past: Luke Giugliano, John Simon Partners: Dr. Jennifer Wilcox
Steam methane reforming (SMR) is an industrial power generation process that converts natural gas to hydrogen, separates the hydrogen through a palladium membrane, and burns it to extract energy. The process has enormous potential for waste heat recovery, CO2 sequestration, and reduction of capital cost. The goal of this study is to investigate the technoeconomic feasibility of an optimized SMR system with CO2 capture relative to other carbon-free power generation technologies. A thermodynamic model was developed to design and analyze chemical reactions, power generation, and waste heat recovery. The model is currently being validated using experimental data from a 100 W scale test facility built at Colorado State University. The overall system efficiency is currently at 43% and may be driven higher at elevated pressures.
In this effort, Colorado State University and the Colorado School of Mines are developing an a low-cost system that can simultaneously produce electricity and separate CO2 from natural gas at scales relevant for distributed generation. The team is (1) developing a detailed cost estimate for a system at the 50 kWe scale, and (2) documenting heat integration concepts. Since the beginning of 2017, the team has begun designing and fabricating a system at a 100 W scale for proof of concept demonstration. Test results from this facility will be used to guide the estimate of a scaled up version. 6 MWe is a relevant scale for economic estimation, and thus the team has begun developing various heat integration concepts for the system and the membrane reformer.
Waste Heat Availability from a High Temperature Diesel Engine
Past: Jonas Adler
Manufacturers continue to make incremental gains in internal combustion engine efficiency, but as much as 60% of the fuel energy may still be rejected to the environment as waste heat. Research on recovering this waste heat has produced disappointing results due to the low heat capacity rate of the exhaust gases and the low temperature of the coolant (~90°C), which limit the average relative efficiency gains to less than 10%. Increasing the engine coolant temperature would greatly increase the availability of waste heat and could result in efficiency gains of over 20%. A small diesel engine test cell was built and tested to measure the availability of waste heat as the engine coolant temperature is incrementally increased. An energy balance was performed on the modified Daihatsu three-cylinder diesel engine, which uses a copper head gasket, upgraded engine oil seals, and custom oil and cooling systems. The results from the experiment were used to model the output of a waste heat recovery system and demonstrate the possible efficiency gains.
Multi-functional Electrolyte for Li-Ion Batteries
Past: Kevin Westhoff PARTNERS: PRIETO BATTERY
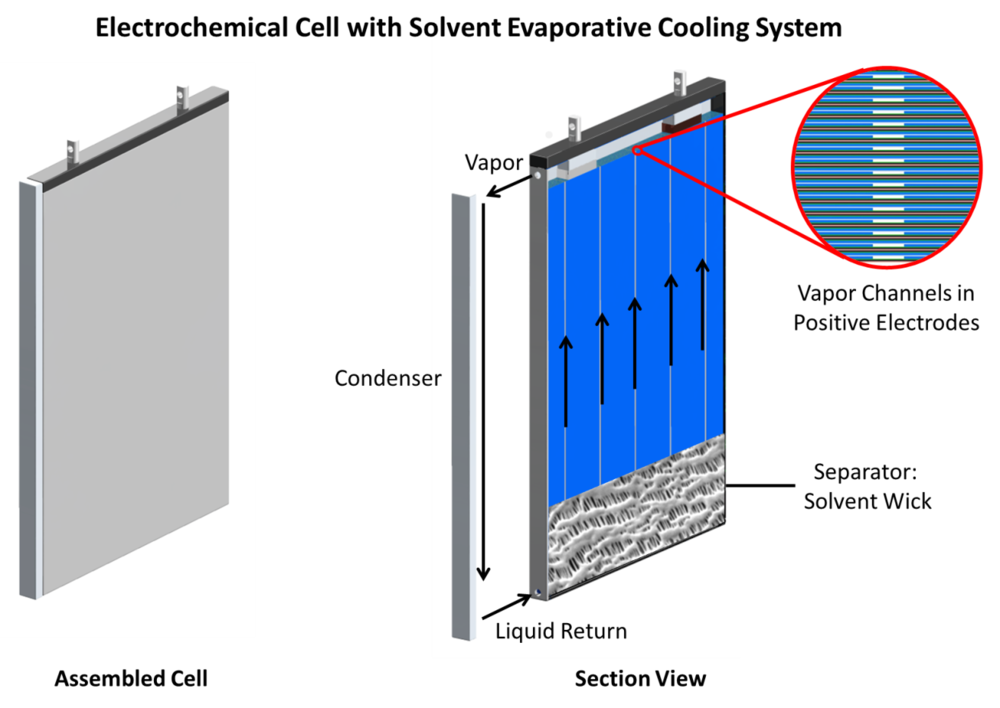
The high thermal conduction resistances of lithium-ion batteries severely limits the effectiveness of conventional external thermal management systems. To remove heat from the insulated interior portions of the cell, a large temperature gradient is required across the cell, and the center of the electrode stack can exceed the thermal runaway onset temperature even under normal cycling conditions. One potential solution is to remove heat locally inside the cell by evaporating a volatile component of the electrolyte. In this system, a high vapor pressure co-solvent evaporates at low temperature prior to triggering thermal runaway. The vapor generated is transported to the skin of the cell, where it is condensed and transported back to the internal portion of the cell via surface tension forces. For this system to function, a co-solvent that has a boiling point below the thermal runaway onset temperature must also allow the cell to function under normal operating conditions. Low boiling point hydrofluoroethers (HFE) were first used by Arai to reduce LIB electrolyte flash points, and have been proven to be compatible with LIB chemistry. In the present study, HFE-7000 and ethyl methyl carbonate (EMC) are used to solvate 1.0 M LiTFSI to produce a candidate electrolyte for the proposed cooling system. Lithium titanate oxide (Li4Ti5O12), copper antimonide (Cu2Sb), and lithium iron phosphate (LiFePO4) are used in half and full cells with the candidate electrolyte for cycling and electrochemical impedance spectroscopy tests, and testing results show similar performance characteristics as compared with a conventional carbonate-only electrolyte (1.0 M LiPF6 in 3:7 ethylene carbonate/diethyl carbonate). The same battery active materials are evaluated in a custom electrolyte boiling facility to evaluate electrochemical performance, and test results show that full electrochemical cells operate similarly even when a portion of the more volatile HFE-7000 is continuously evaporated.
Physical and Electochemical Parameter Measurements of Commercial Lithium-Ion Cells
Past: Kevin Westhoff, Trevor Vernon PARTNERS: EVERETT JACKSON, DR AMY PRIETO, DR GREGORY PLETT
Five commercial lithium-ion batteries (LIB) were disassembled and analyzed for specific physical and electrochemical parameters. Cell disassembly was completed in an Argon glove box. Detailed measurements were performed on the physical construction of the batteries with the goal of determining the volume of active material in each electrode. The open circuit potential (OCP) as a function of temperature and state of charge (SOC) for the cathode and anode active materials versus Li/Li+ was measured. These tests used ¾” half-cells cycled at a C/30 rate on an Arbin battery tester while under strict environmental temperature control. The diffusion coefficient of Li+ as a function of temperature and SOC in the active materials was measured using the Galvanostatic Intermittent Titration Technique (GITT). Nuclear Magnetic Resonance Spectroscopy (NMR) was used to determine the composition of the electrolyte, if enough of an electrolyte sample was harvested during cell disassembly. Extensive Scanning Electron Microscope (SEM) imaging was performed on the electrodes to determine the coating thickness and active material particle size. Additionally, Energy Dispersive Spectroscopy (EDS) was performed on all electrode samples to determine the elemental composition of the active materials. This data was then coupled with X-Ray Diffraction (XRD) crystallographic data to produce an equivalent unit cell of the active material in order to determine the theoretical lithium storage capacity. The composite electrode electronic conductivity was also determined as a function of temperature with a Gamry Reference 3000 Potentiostat.